 Their oxidation number, therefore is -1 and the charge on both atoms is 1. The latter consist of two sulphur atoms joined by a covalent bond. 
Pyrite and marcasite consist of Fe2+ cations and disulphide anions. perchlorate, periodate, persulphate, etc.). Though, most chemists would believe that a peroxosulphide is meant, for the prefix "per-" always denotes oxygen-rich compounds (e.g. The term "persulphide"is not a defined compound name. Recent Images in DiscussionsĢ1st Jul 2006 13:09 UTC Peter Haas To follow up on nomenclature, the disulphide belongs to the group of polysulphides. Currier Digital LibraryOpen discussion area. Techniques for CollectorsOpen discussion area. Minerals and MuseumsOpen discussion area. Mineralogical ClassificationOpen discussion area. Lost and Stolen SpecimensOpen discussion area. Therefore, the Roman numeral (I) must be used to show the charge of the copper ion.╳Discussions □ Home □ Search □ Latest Groups EducationOpen discussion area.The table below shows that 2 Cu + will balance the charge of 1 S 2.For example, Cu, can have a charge of 1+ or 2+.To find the Roman numeral for the element, first refer to the periodic table. The transition metals below do not need a Roman numeral in the names of their compounds because they only form one ion. 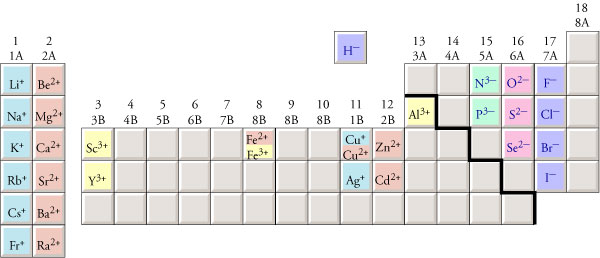
For example, Fe forms Fe 2+ or Fe 3+ ions in compounds. Roman numerals are only used for transition metals that have more than one ion. Transition metals that do not require Roman numerals So, the compound is silver(I) chloride, but it is commonly known as silver chloride.Refer to the periodic table above, Ag forms 1+ charge and Cl forms 1- charge.It falls in the category of ionic compound because it has a metal (Ag) bonded to a non-metal (Cl).First, identify the type of compound it is.What is the formula of the compound AgCl? The name of the compound is Lead(II) oxide since lead has a charge of 2+.The compound name is: Iron(III) Chloride.To name the compound, we must use roman numerals.We need three Cl – anions to balance the charge of one Fe 3+cation. If the iron 3+ ion combines with chloride, what is the formula of the compound? The compound name is: Iron(II) Chloride.Fe is a transition metal, we must use a roman numeral to indicate the charge. 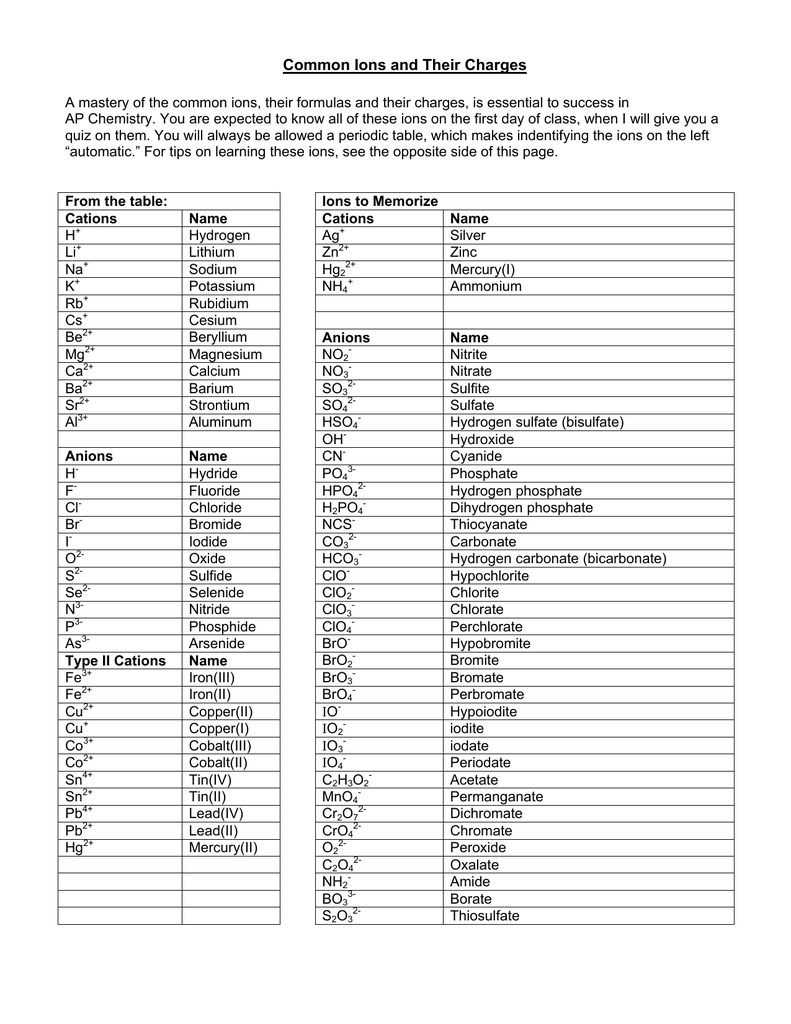
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
